Red Blood Cell RBC
Description
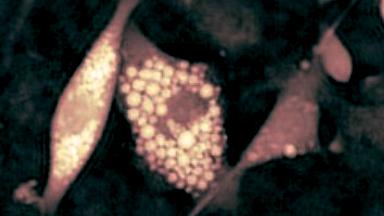
The RBC solution by DHM® is used to measure clinically relevant parameters such as Hemoglobin Content and Mean Cell Volume (MCV) on intact individual RBCs. The results are compared with values achieved by conventional techniques on RBC from the same donor and with previously published figures.
The mean cell volume (MCV), red blood cell distribution width (RDW), and mean cell hemoglobin concentration (MCHC) measurements were also validated using an impedance volume analyzer.
Material and methods
- Cells: 100–150 microliters of blood were drawn from healthy donors (fingerpick). Blood cells were sedimented, buffy coat removed. RBCs were washed and introduced into the experimental perfusion chamber
- DHM® solution: RBC
- Method: Quality assessment by Impedance Volume Analyzer
Results
The hemoglobin content measurements obtained with the DHM® are in excellent agreement with those obtained with the impedance volume analyzer.
The Sysmex KX-21 compares the optical density of lysed erythrocytes versus a calibration curve, allows to obtain a very precise measurement on cell populations and provides high throughput, but is highly invasive as cell lysis is required.
On the contrary, DHM® measures the hemoglobin concentration of individual cells in a manner that is independent of the shape and state of the cell. The ability of DHM® to measure the hematological parameters of erythrocytes independently and accurately may allow objective evaluation of the biological variation in many RBC disorders in a non-invasive and label-free manner.
Publication
Description
The RBC solution by DHM® is used to measure morphology, volume, refractive index (related to hemoglobin content) on intact individual RBCs. The results are compared with values achieved by conventional techniques on RBC from the same donor and with previously published figures
Material and methods
- Cells: 100–150 microliters of blood were drawn from healthy donors fingerpick, collected, and diluted. Blood cells were sedimented, buffy coat removed. RBCs were washed and introduced into the experimental perfusion chamber
- DHM® solution: RBC
- Method: Quality assessment by laser confocal microscopy and by Impedance Volume Analyzer
Results
The hemoglobin content measurements obtained with the DHM® are in good agreement with those obtained with the laser confocal microscope and impedance volume analyzer.
The DHM® measurements are label-free and non-invasive. DHM® enables to measure single RBC as well as to make statistical analysis over many cells, whereas the long measurement procedure of CSLM makes statistical analysis difficult, and impedance volume analyzer does not enable to measure single cells. The ability of DHM® to measure the hematological parameters of erythrocytes independently and accurately may allow objective evaluation of the biological variation in many RBC disorders.
Description
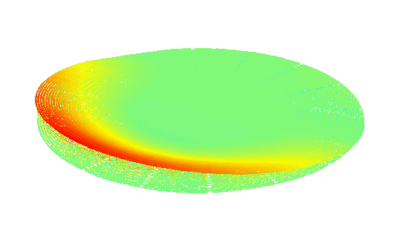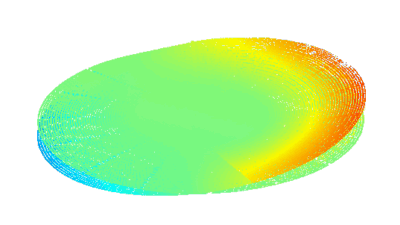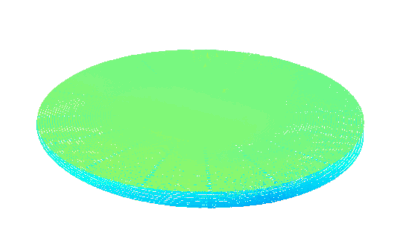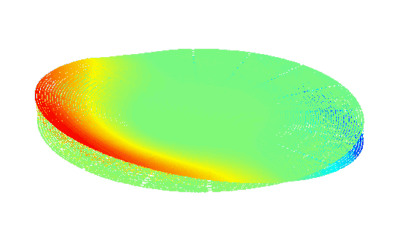
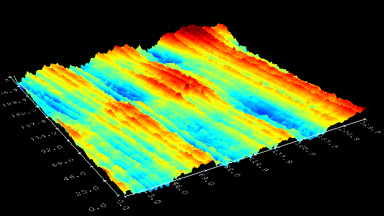
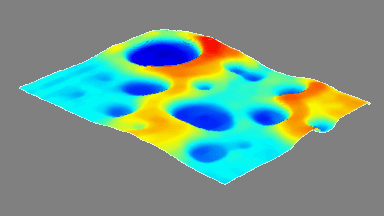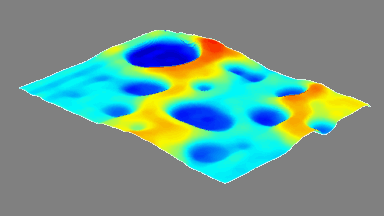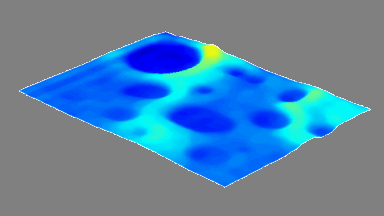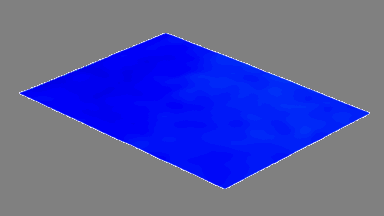
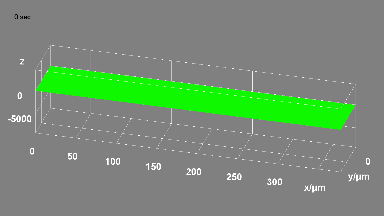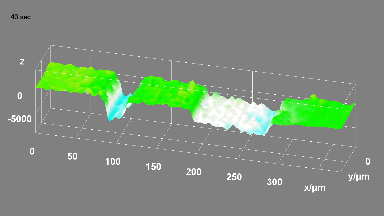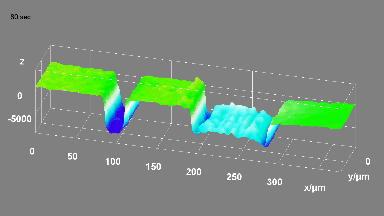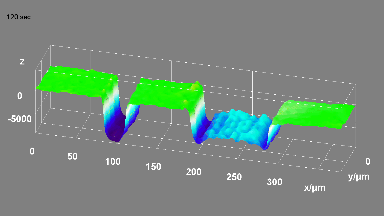
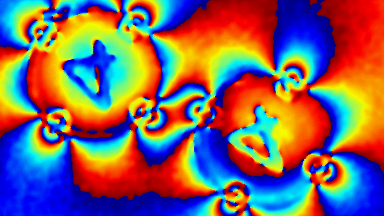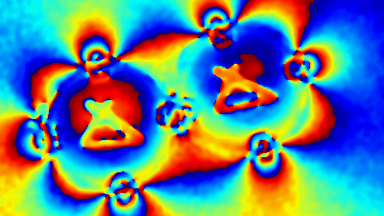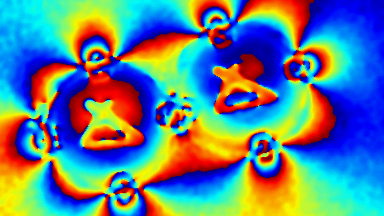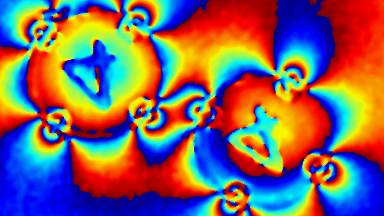
Cell flickering is a good indicator of RBC health. DHM® is used for spatial analysis of red blood Cell Membrane Fluctuations (CMF). CMF have been the subject of many studies in order to obtain a better understanding of these remarkable biomechanical membrane properties altered by some pathological states including blood diseases. In particular the discussion over the thermal or metabolic origin of the CMF has led in the past to a large number of investigations and modelings.
This application presents an analysis of the CMF of RBCs by measuring their time sequence by DHM, extracting the membrane fluctuation amplitude, and analyzing cell membrane topology with an orthogonal subspace decomposition of the data, identified and quantified as the eigenmode basis of CMF that minimizes the deformation energy of the RBC structure.
Material and methods
- Cells: 100–150 microliters of blood were drawn from healthy donors fingerpick, collected, and diluted. Blood cells were sedimented, buffy coat removed. RBCs were washed and introduced into the experimental perfusion chamber
- DHM® solution: RBC
- Method: Data: time sequences acquired by DHM®. Analysis: membrane fluctuation amplitude and analysis extracted from cell membrane topology
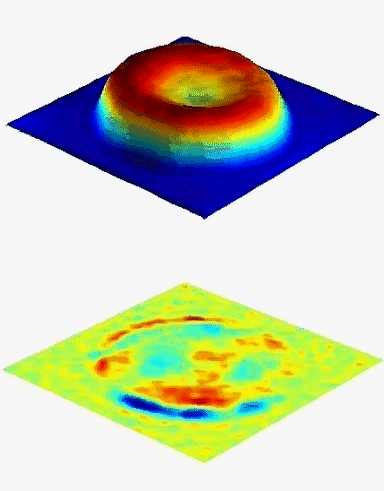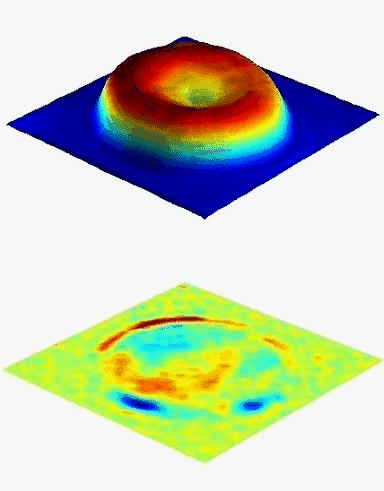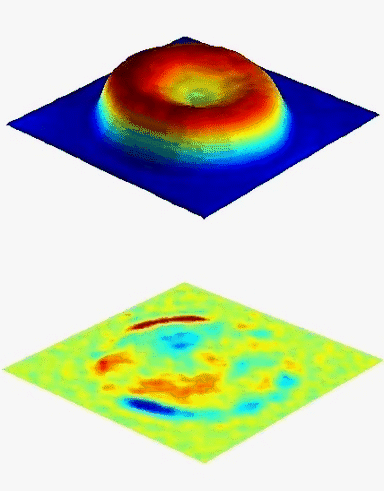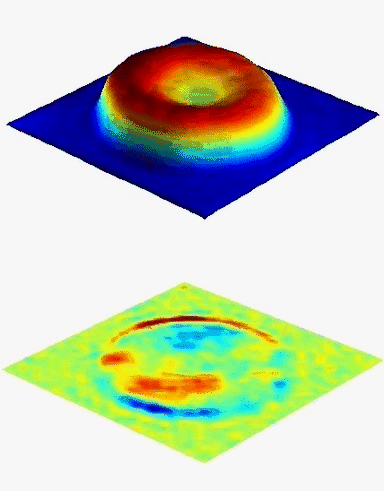
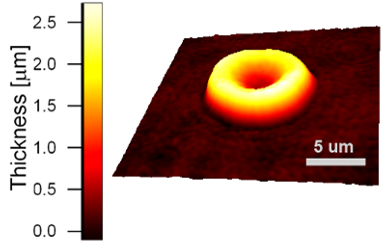
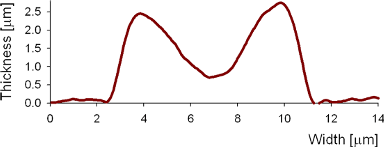
Results
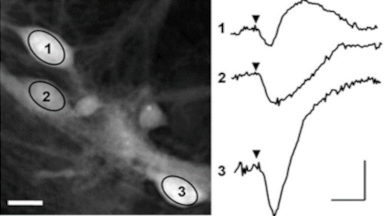
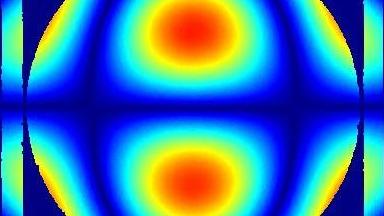
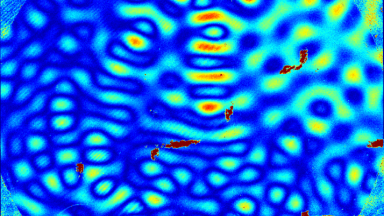
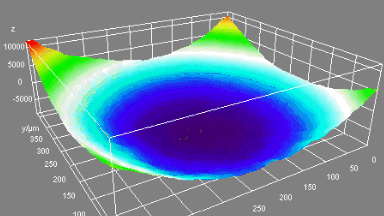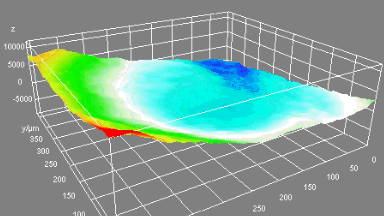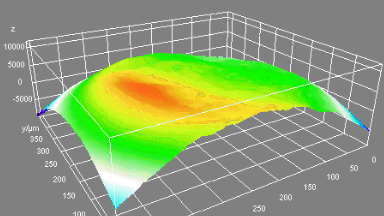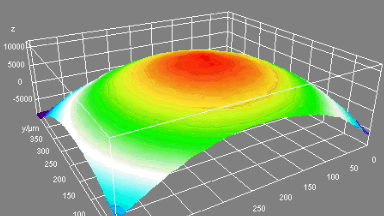
Uneven distribution of CMF amplitudes over the RBC surface is observed, showing maximal values in a ring corresponding to the highest points on the RBC torus as well as in some scattered areas in the inner region of the RBC. CMF amplitudes of 35.9±8.9 nm and 4.7±0.5 nm (averaged over the cell surface) were determined for normal and ethanol-fixed RBCs, respectively.
Moreover, by fitting the observed CMF modes with a theoretical dynamic model, we find that the CMF are mainly governed by the bending elasticity of the membrane and that shear and tension elasticities have only a marginal influence on the membrane fluctuations of the discocyte RBC. Furthermore, the experiments show that the role of ATP as a driving force of CMF is questionable. ATP, however, seems to be required to maintain the unique biomechanical properties of the RBC membrane that lead to thermally excited CMF.
Publications
- Spatial analysis of erythrocyte membrane fluctuations by digital holographic microscopy, B. rappaz, et. al., Blood Cells Mol Dis. 2009 May-Jun; 42(3):228-32
- Spatially-Resolved Eigenmode Decomposition of Red Blood Cells Membrane Fluctuations Questions the Role of ATP in Flickering, D.Boss, et. al., PLOS One , vol. 7, no. 8, 2012
Description
An increase of oxygen saturation within blood bags and metabolic dysregulation occur during storage of red blood cells (RBCs). It leads to the gradual exhaustion of RBC antioxidant protective system and, consequently, to a deleterious state of oxidative stress that plays a major role in the apparition of the so-called storage lesions.
Material and methods
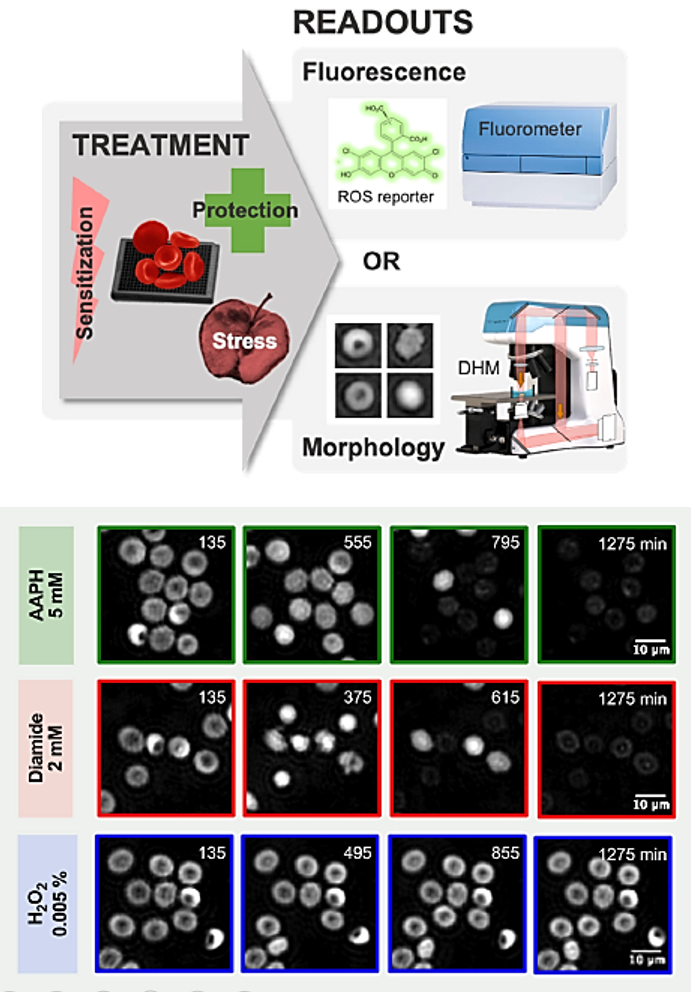
The screening assay was conducted on an automated DHM-T1000 with a motorized stage for High-content/high-throughput screening applications.
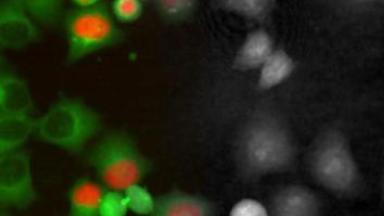
Digital Holographic Microscopy is used to quantify the morphological alterations induced by three oxidants (AAPH, diamide and H2O2) and the protective effect of four antioxidants molecules (ascorbic acid, uric acid, trolox and resveratrol) at different concentrations.
Results
This approach allowed to identify a specif shape of RBC called “Tirocytes” and open the way to large screens of molecules on RBC to improve their storage health.
“Our team has been using digital holographic microscopy, developed by Lyncée Tec, for several years to easily investigate the ex vivo aging of red blood cells in the context of transfusion medicine. The high-throughput capability of DHM is an advantage to study libraries of compounds that can improve the storage of red blood cells. In combination to fluorescence microscopy, this instrument will provide complementary information on cell morphology and oxidative stress.”
Michel Prudent, PhD
Head of Research and Development Products at Transfusion Interrégionale CRS